Our Businesses
- Home
- Corporate Information
- Our Businesses
We provide a wide range of catheters that benefit from the wealth of knowledge on materials that Zeon Corporation possesses as a chemical manufacturer as well as the proprietary material processing technology we have developed over the years in Takaoka.
Cardiovascular Business
Zeon Medical Inc. was founded in 1990 to manufacture and sell the world’s first ventricular assist device. With pride in the company’s origin, we provide a wide range of products such as IABP balloons and associated drive units that assist with blood circulation, fractional flow reserve (FFR) measuring systems for diagnosing the state of the stenosis of coronary arteries, and balloons for percutaneous transluminal coronary angioplasty for treating the stenosis of coronary arteries and improving blood flow. We also handle various catheters for percutaneous coronary intervention.
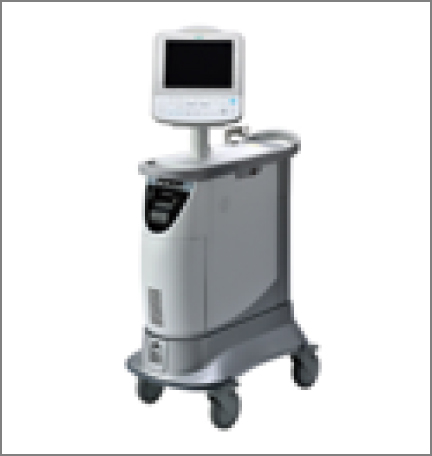
IABP drive unit

FFR
Digestive Medical Devices
We develop and provide unique treatment devices for digestive organs. These reduce patient discomfort and leverage our proprietary advanced processing technology with full consideration for the opinions of medical professionals on these devices.
Treatment Devices for Bile Duct Diseases
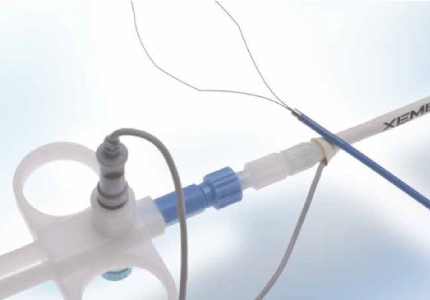
We are developing and providing treatment devices for ERCP to diagnose and treat bile and pancreatic duct diseases. Moreover, we provide treatment devices for a wide range of indications, such as bile duct stents that improve their stricture, baskets and crushers to remove gallstones, and balloon catheters.

Basket

Stent
Treatment of Digestive Diseases
Recent advances in diagnostic and treatment technologies have made possible the early detection of cancers. Consequently, endoscopic treatment has drawn increased attention, as it allows for shorter hospital stays and less of a burden on patients compared to surgery. We develop proprietary devices for endoscopic treatment suitable for specific cancer stages and symptoms, thereby contributing to improvements in medical technology.
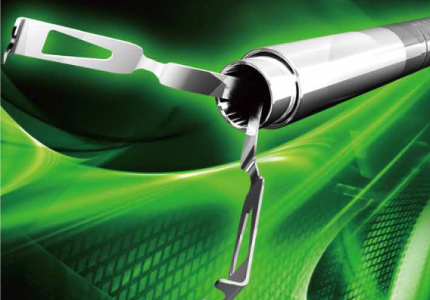
Hemostatic clip
Snare
Main Business Sites
Head Office
The head office was relocated to the Shin-Marunouchi Center Building near the Marunouchi North Exit of Tokyo Station in August 2017. Based at the head office, domestic and overseas sales, marketing, regulatory affairs, logistics, R&D, and administration collaborate on a wide range of activities, from daily business operations to the review of medium- to long-term business strategies.

Manufacturing Plant・Technology Development
Under the motto, “Utilizing Japanese technologies to manufacture high-quality products,” We continue to produce and improve products that contribute to improvement. And we have been driving the evolution of our manufacturing process with a focus on increasing productivity, reducing costs, and developing human resources.


